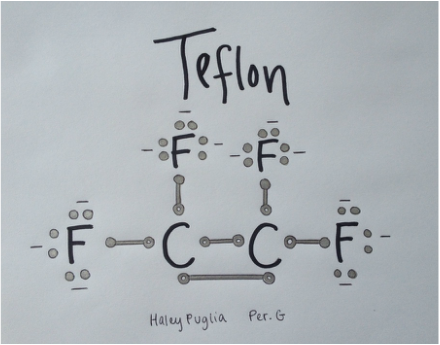
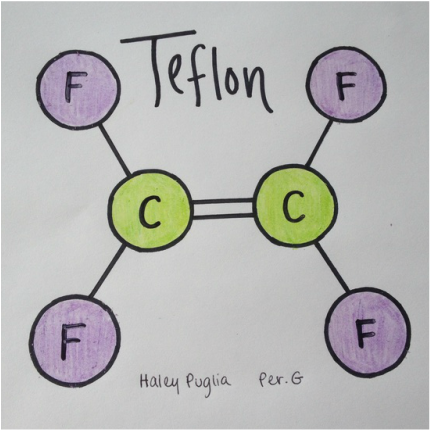
Structural FormulaLewis StructureTo the right is the Lewis Structure of Teflon. Because the molecular formula is C2F4 there are two Carbons (C) and four Fluorine (F). There is a double bond between the two Carbons and the bonds between Carbon and Fluorine are single. The circles around the elements represent the electrons that the element posses. Each Carbon has four electrons where each Fluorine has seven. The minus signs next to the electrons represent negativity. The negativity of the molecule is spread evenly throughout making it non-polar. This means that there are not both positive and negative sides to the molecule. It is instead asymmetrical.
|
|